|
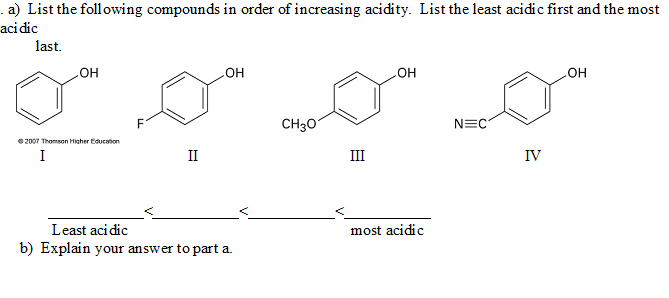
This point causes a lot of confusion due to the presence of two seemingly conflicting trends. While far from impossible, forming the dianion can be difficult due to the buildup of negative charge and the corresponding electronic repulsions that result. Note that once a conjugate base (B-) is negative, a second deprotonation will make the dianion (B 2-). This is, of course, easiest to do when an atom bears a charge of +1 in the first place, and becomes progressively more difficult as the overall charge becomes negative. Removal of a proton, H+, decreases the formal charge on an atom or molecule by one unit. With that out of the way, let’s get started. 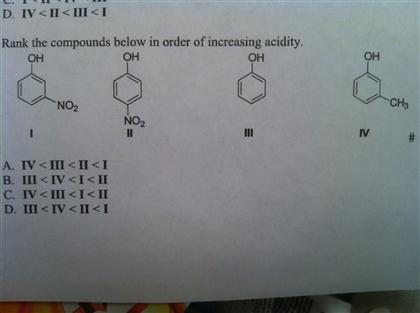
Put another way, strong acids have weak conjugate bases, and vice versa. 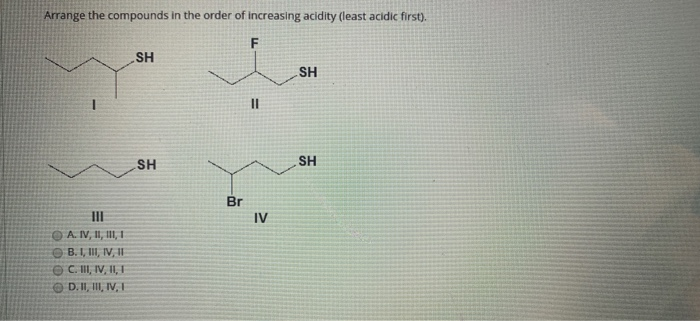
Therefore pK a is also a measure of how stable the conjugate base is.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
